You may not expect pharmaceutical manufacturing and facility maintenance in a commercial or residential building to have much in common, but they’re more related than you think. Not only do engineers in both often deal with similar types of equipment – pumps, valves, filters, heating, cooling, etc., they are often finding creative solutions to unexpected challenges.
I’ve also seen teams in both industries benefit tremendously when they are well trained and really care about the people, equipment, and process they impact.
How mentorship saved my team
Tony and Gary (a different Gary) are engineers from the last biopharma plant I worked in, and a great example of strong mentors. The wisdom they shared once helped our team avoid a costly schedule delay to a multimillion dollar process.
Here’s what happened:
We were commissioning a new processing step for a drug in clinical trials. The liquid being processed came from a large fermenter, like what’s used when brewing beer. The fermenter grows cells that produce a protein. The protein in the liquid was the active drug substance that we needed to isolate. The process was a simple filtration followed by a specialized filtration.
Doing so required three steps –
First step: Pass the mixture through dead end, size exclusion filters.
This is the way most filters work:
A filter with pores slightly larger than the protein is selected. As the liquid passes through the filter, large waste material gets trapped while the protein passes through and is retained.
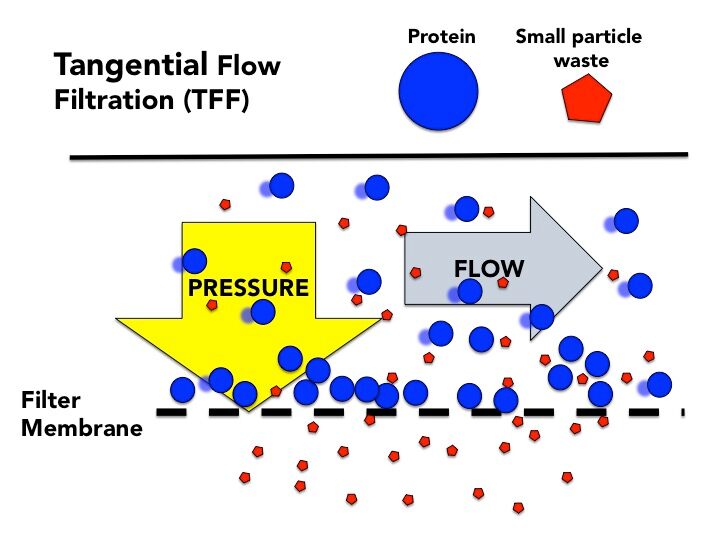
Second step: Remove smaller particles with a tangential flow filter.
Tangential flow filtration (TFF), sometimes called cross flow, is a bit different. Liquid flows across a filter membrane, tangent to the membrane’s surface. The pores on this filter membrane are slightly smaller than the protein so it doesn’t pass through. And since it’s flowing across the membrane, it doesn’t get stuck, it bounces by, returning to the tank it started in.
The protein solution continues recirculating and pressure is applied. The pressure forces the small particles through the membrane as waste. The protein is retained, the volume reduced, and the small waste is forced out.
Third step: Introduce a storage solution and freeze it.
When the volume in step 2 reaches a certain level, a new storage solution is mixed with the recirculating liquid. The solution is added at the same rate that the waste is being removed. Enough storage solution is added so the fluid originally mixed with the protein gets replaced. Finally the mixture, now made up of only the protein and storage solution, is frozen and stored.
At least that’s how it’s supposed to happen…but we had a problem, and the clock was ticking.
The storage solution flow rate wasn’t high enough. It was causing us to run the process too slowly. Running slower reduced the pressure and flow rate across the filter membrane. TFF filters only work properly when run at a specified pressure and flowrate.
This had potential to cause delays. In this business, delays could cost hundreds of thousands of dollars. But we had skills. Tony had always been meticulous about explaining the important details that weren’t in the training manual.
His knowledge went far beyond what standard training provided. He was old school and he liked sharing his expertise. Without people like Tony, I may have hit the stop button and found something else to do while an investigation was initiated.
Instead, my team and I first tested the peristaltic pumps responsible for delivering the storage solution. I knew a lot about these pumps and the concept behind using them because of people like Tony. We confidently eliminated them as the root cause.
Then we took a close look at the tubing that fit into the pumps. This was a disposable item. Gary knew everything about disposable parts. He had taken the time to explain the behind the scenes system of having these parts manufactured. He showed me that there were always two manufacturers: one to back up the other, in case one company were to go out of business or something.
I checked it out. Sure enough one supplier made their tubing out of rubber that had a slightly different wall thickness and density. Technically the tubing met the specifications, but the difference could affect the performance. With this new information, we started testing.
With a bit more bureaucracy than I’m indicating here, the problem was verified. Sure enough, that was it! We changed tubing and flow rate increased to appropriate levels.
Sharing knowledge makes all the difference
Would the problem have been identified in an investigation? Probably, but we were able to skip that step. We saved a bunch of time and money because of people like Tony and Gary. They chose to share their knowledge and experience, and it made all the difference.
Training & Mentorship
Pharmaceutical manufacturing uses written standard operating procedures (SOPs). Almost every task and training must be documented for any procedures you perform, including how to write SOPs.
This is incredibly valuable. But, it’s not all there is to know. Only by spending time and learning directly from Tony and Gary did I truly understand our systems well enough to make a save like this one.
Mentors, like those involved with the pump tubing incident, have taught me (and many others) valuable things over the years:
- How to troubleshoot and predict breakdown or failure
- Why specific processes or materials are selected
- To read technical drawings and sketches
- How to deal with vendors and connect with experts
- To promote knowledge as an advantage
- To gain experience for career advancement
- About more diverse career paths
Takeaways
A funny thing about pharma manufacturing is that not long after this, the drug failed in clinical trials. The plug was pulled on this project, and all this work was filed away.
Was it all a waste of time? I don’t think so. The skills and learning that saved the schedule that day will prevent loss again. The people on this team became more valuable and it was all made possible by people like Tony and Gary who wanted us to learn.
Knowledge in the hands of many is powerful. Not sharing unique or advanced experience is a mistake.
If you have some uncommon knowledge like Gary or Tony did, find a way to share it with the people around you. You’ll be kicking yourself if you lose good employees for better learning opportunities.
And if you don’t know something, find a way to learn it, especially from those with more experience. Bosses love that stuff. Ask those around you to show you what he or she knows, and don’t miss an opportunity to learn from a mentor.
You never know when that random piece of knowledge could save the day.
LogCheck’s digital logbook makes it easy to spread knowledge through your team. Whether you need to know what happened in the field today or you want to share notes so they’re always on hand, LogCheck helps keep everyone informed. Click the button below to sign up for a trial to see for yourself:
Try LogCheck today!



0 Comments